 
The sum of the mass numbers of the reactants equals the sum of the mass numbers of the products.Nuclear reactions also follow conservation laws, and they are balanced in two ways: A balanced nuclear reaction equation indicates that there is a rearrangement during a nuclear reaction, but of nucleons (subatomic particles within the atoms’ nuclei) rather than atoms. Balancing Nuclear ReactionsĪ balanced chemical reaction equation reflects the fact that during a chemical reaction, bonds break and form, and atoms are rearranged, but the total numbers of atoms of each element are conserved and do not change. Due to the much larger energy differences between nuclear energy shells, gamma rays emanating from a nucleus have energies that are typically millions of times larger than electromagnetic radiation emanating from electronic transitions. Gamma rays are a type of high energy electromagnetic radiation produced when a nucleus undergoes a transition from a higher to a lower energy state, similar to how a photon is produced by an electronic transition from a higher to a lower energy level. −1 0 e + +1 0 e ⟶ γ + γ −1 0 e + +1 0 e ⟶ γ + γĪs seen in the chapter discussing light and electromagnetic radiation, gamma rays compose short wavelength, high-energy electromagnetic radiation and are (much) more energetic than better-known X-rays that can behave as particles in the wave-particle duality sense. This works because, in general, the ion charge is not important in the balancing of nuclear equations. For example, an alpha particle is a helium nucleus (He) with a charge of +2 and a mass number of 4, so it is symbolized 2 4 He. 
The subscripts and superscripts are necessary for balancing nuclear equations, but are usually optional in other circumstances. 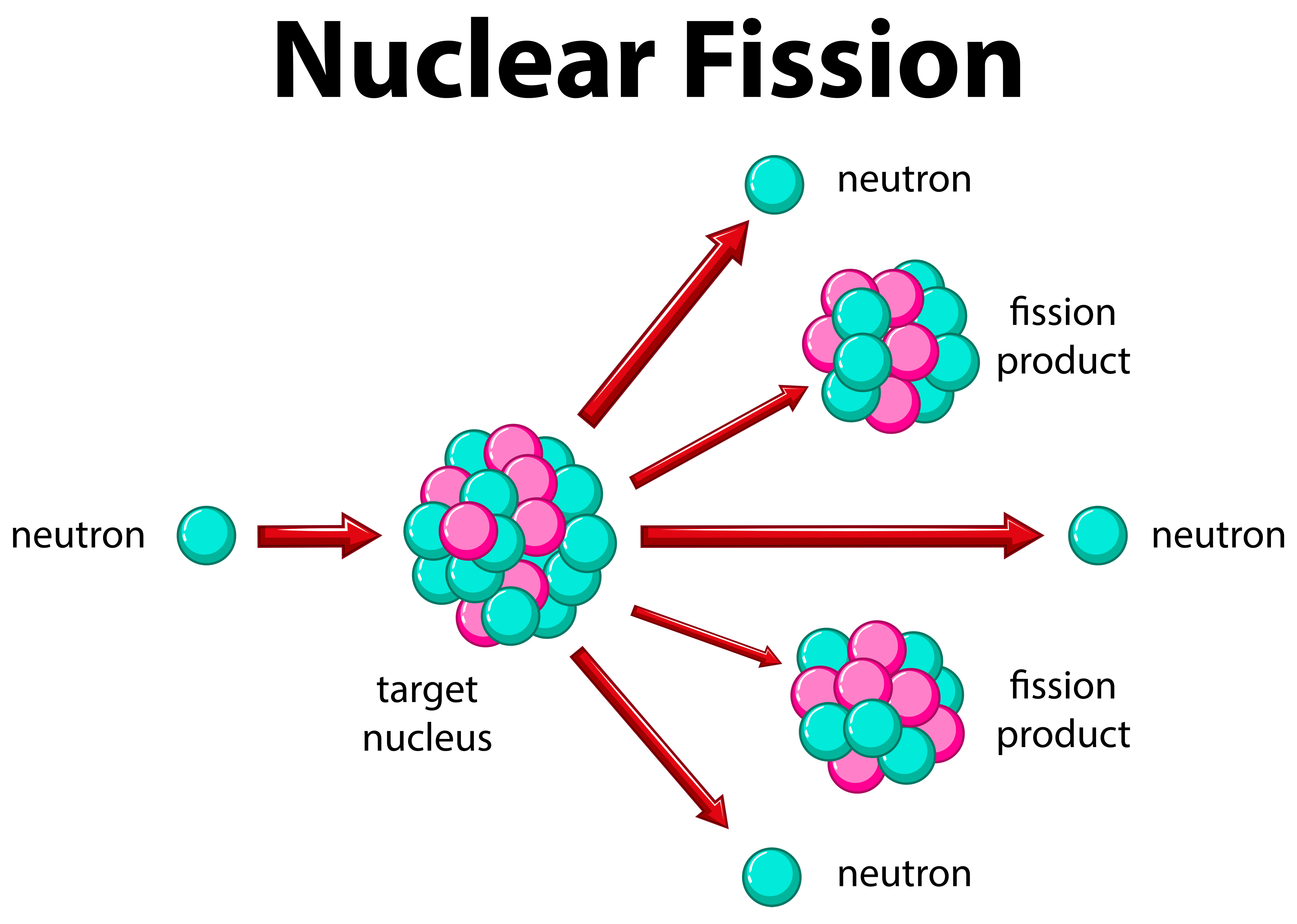
Positrons ( +1 0 e, ( +1 0 e, also represented by the symbol +1 0 β ) +1 0 β ) are positively charged electrons (“anti-electrons”). Beta particles ( −1 0 β, ( −1 0 β, also represented by the symbol −1 0 e ) −1 0 e ) are high-energy electrons, and gamma rays are photons of very high-energy electromagnetic radiation. Alpha particles ( 2 4 He, ( 2 4 He, also represented by the symbol 2 4 α ) 2 4 α ) are high-energy helium nuclei. Protons ( 1 1 p, ( 1 1 p, also represented by the symbol 1 1 H ) 1 1 H ) and neutrons ( 0 1 n ) ( 0 1 n ) are the constituents of atomic nuclei, and have been described previously. The most common are protons, neutrons, alpha particles, beta particles, positrons, and gamma rays, as shown in Figure 21.4. Many entities can be involved in nuclear reactions. To describe a nuclear reaction, we use an equation that identifies the nuclides involved in the reaction, their mass numbers and atomic numbers, and the other particles involved in the reaction. Identify common particles and energies involved in nuclear reactionsĬhanges of nuclei that result in changes in their atomic numbers, mass numbers, or energy states are nuclear reactions.While uranium-235 is the isotope that undergoes fission it is worth noting that uranium-238 atoms can absorb neutrons to become plutonium-239 which is another atom that can undergo fission.By the end of this section, you will be able to: We will consider where the energy comes from in the next section. It is 50 million times more energy than burning the equivalent amount of carbon. The energy released per fission is relatively large.The neutrons may need to be slowed down and are then referred to as thermal neutrons. Both the number and speed of the neutrons is crucial within a working reactor. For fission to occur the neutrons must be going at the right speed – too fast and they will bounce off rather than be absorbed.Once started, fission can become self-sustaining – this is called a chain reaction. The fact that neutrons are also produced means that these neutrons can go on to induce further fissions.These by-products of nuclear power form the majority of the radioactive waste that we will consider next week. Fission products tend to be radioactive. /fission-of-a-uranium-nucleus-141483757-579266b03df78c173498d794.jpg)
In words this would be: ‘A uranium-235 atom absorbs a neutron to become uranium-236 which then undergoes fission to form the products xenon-140 and strontium-93 with three neutrons.’ An equation representing this particular fission would be: The fission products themselves can vary but examples would be xenon-140 and strontium-93. Neutrons are shown as using the same notation as for isotopes. This image shoes the particles and types of radiation involved in fission Figure 14 The particles and types of radiation involved in fission 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
